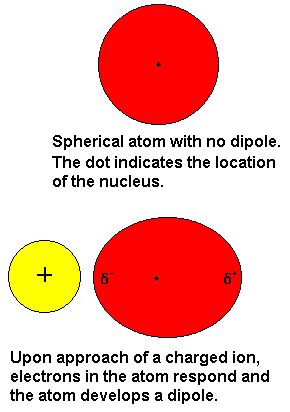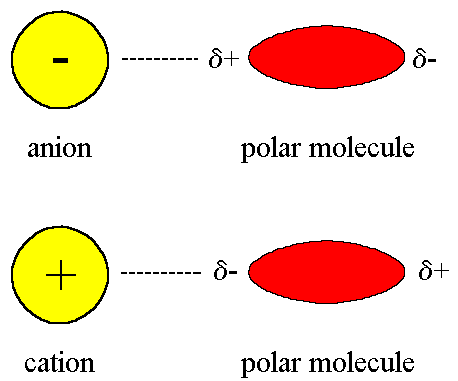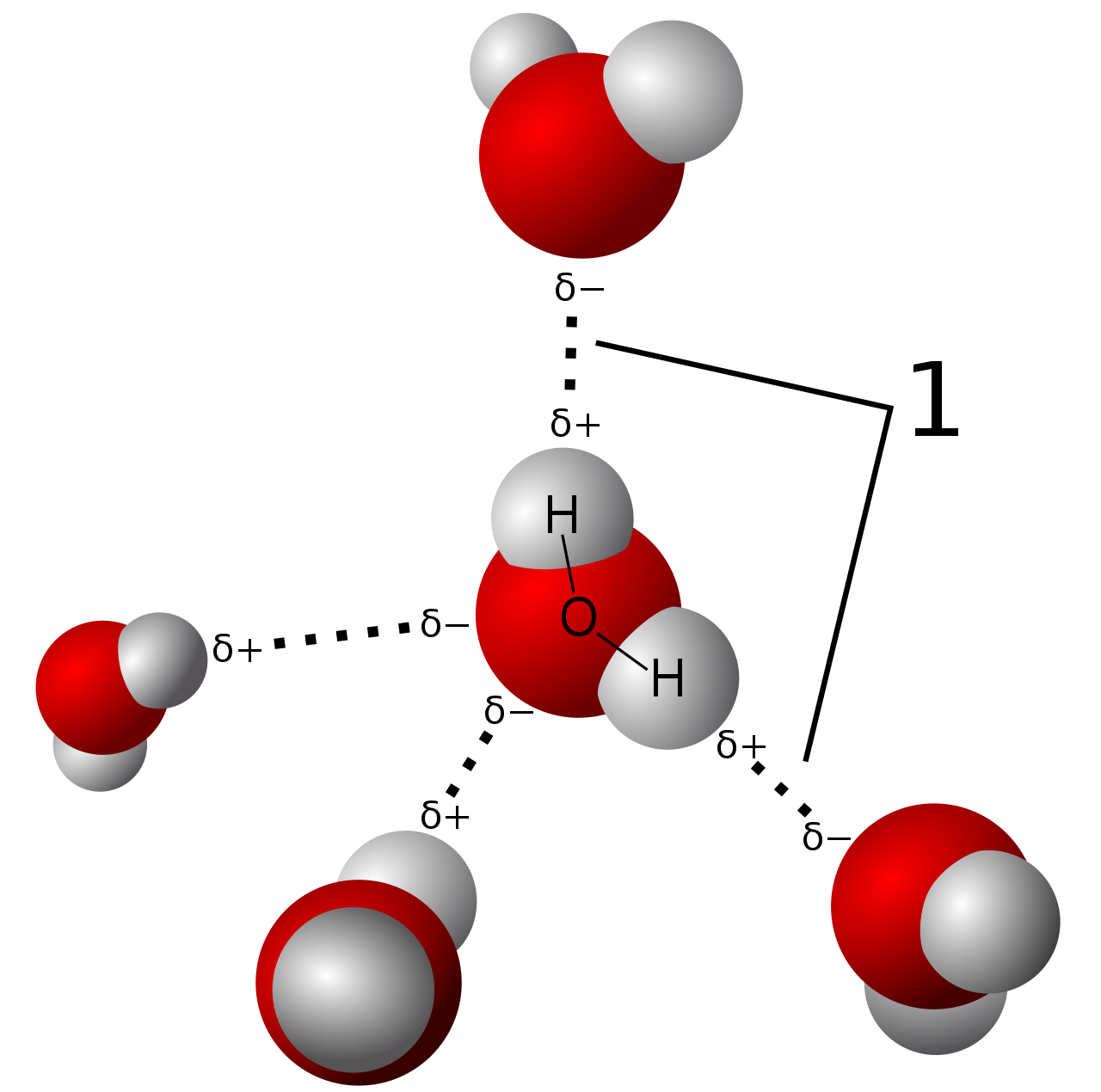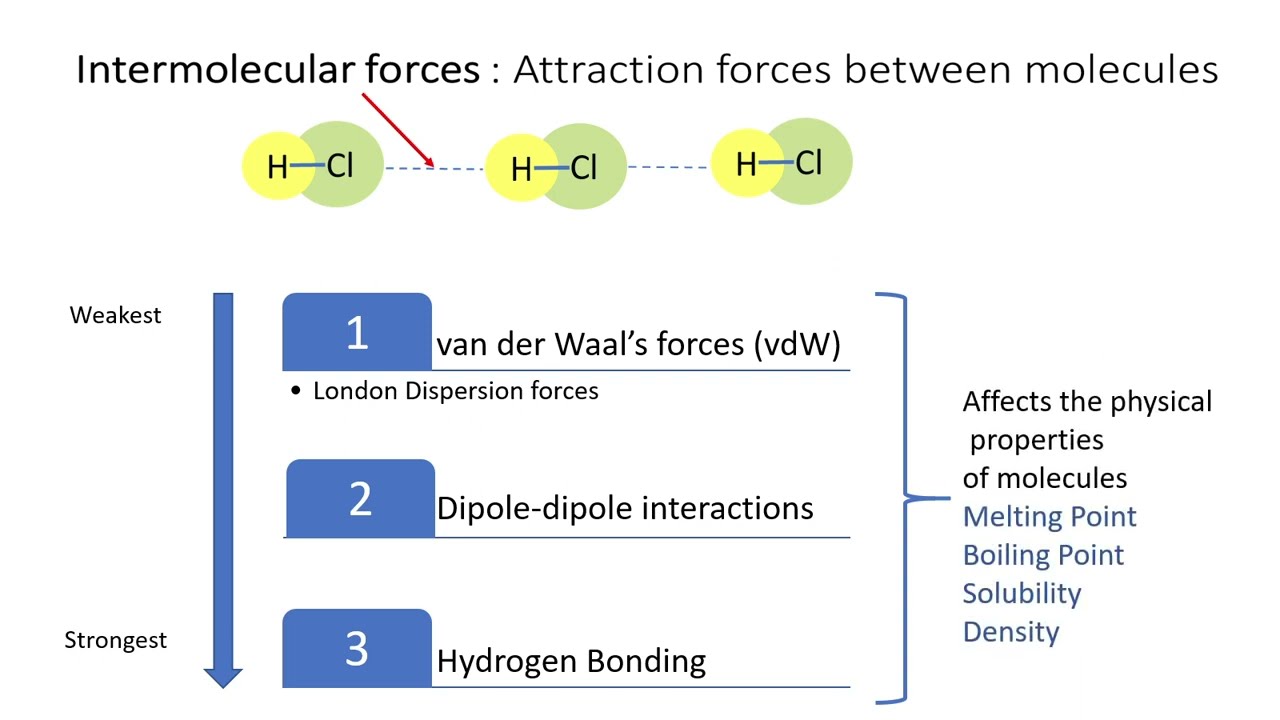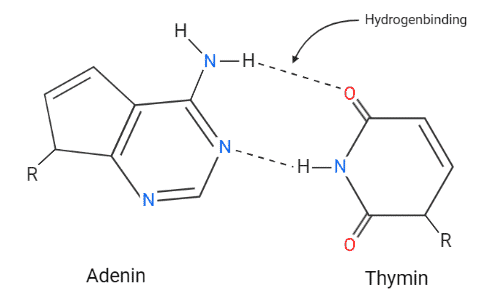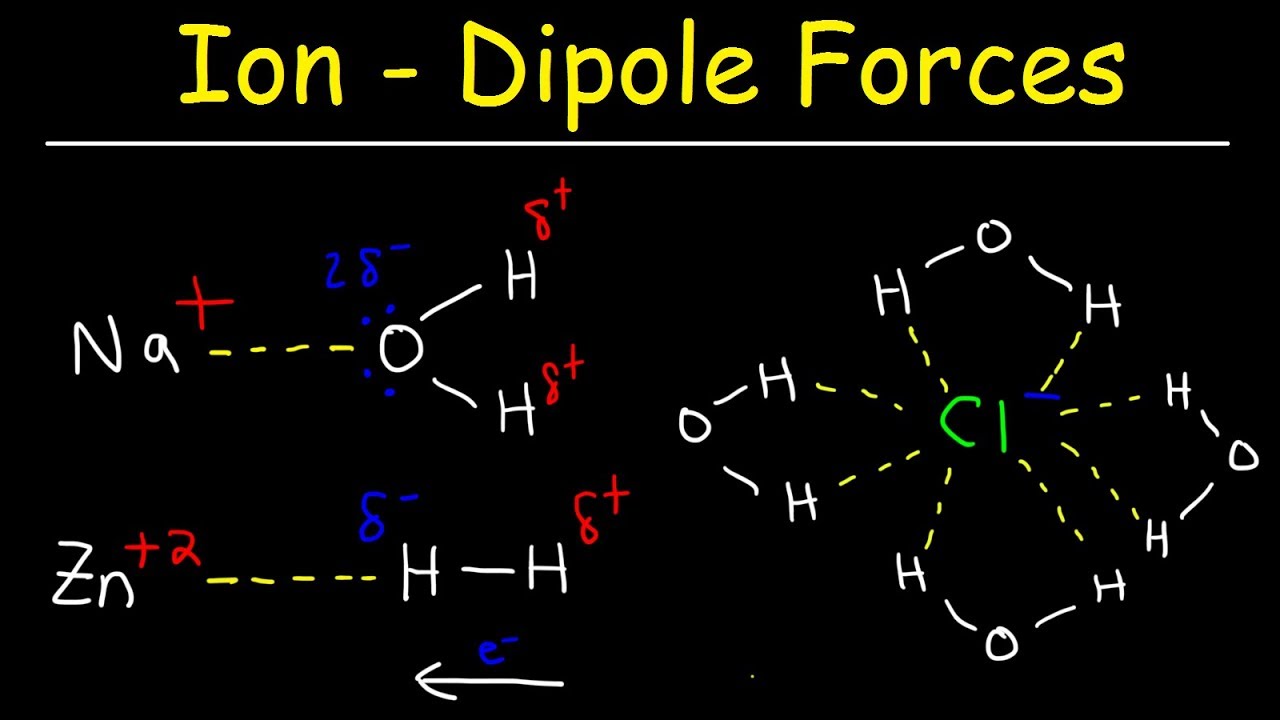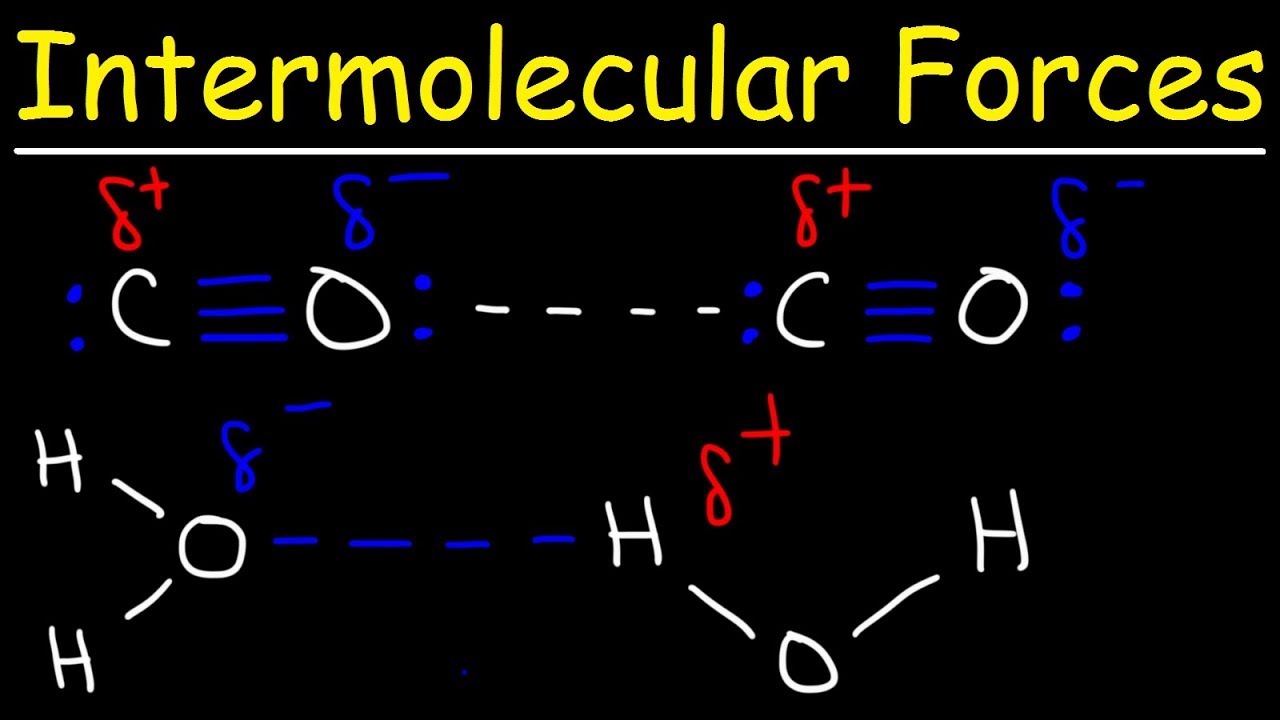
Intermolecular Forces - Hydrogen Bonding, Dipole Dipole Interactions - Boiling Point & Solubility - YouTube

Hydrogen Bonding, Dipole-Dipole & Ion-Dipole Forces: Strong Intermolecular Forces - Video & Lesson Transcript | Study.com

Hydrogen Bonding, Dipole-Dipole & Ion-Dipole Forces: Strong Intermolecular Forces - Video & Lesson Transcript | Study.com

Which of the following are not soluble in water? a. ionic compounds like salt b. polar compounds like sugar c. nonpolar substances like oils | Homework.Study.com
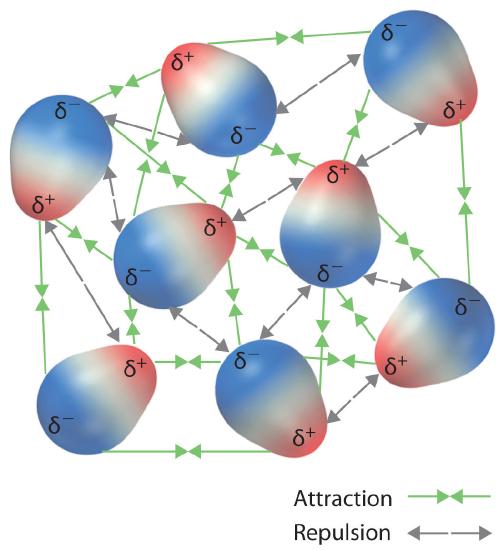
12.6: Intermolecular Forces: Dispersion, Dipole–Dipole, Hydrogen Bonding, and Ion-Dipole - Chemistry LibreTexts
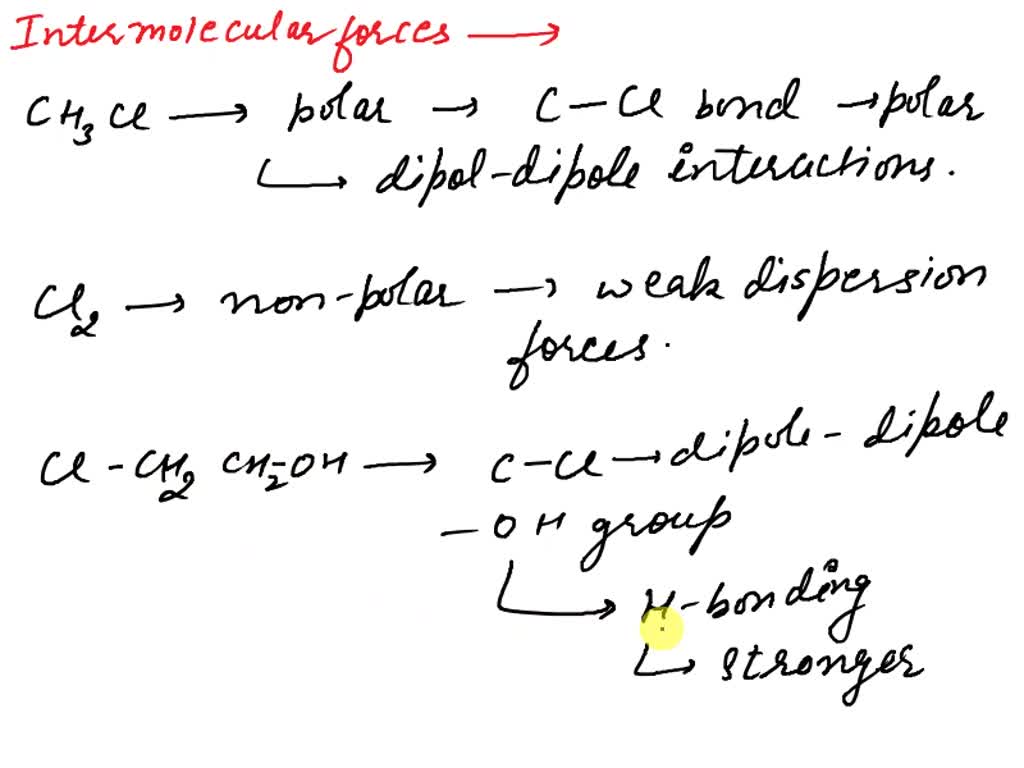
SOLVED: Choose the strongest non-covalent interaction that exists between the following pairs of compounds. BF4- and CHCl3 H2O and SF6 CHCl3 and CH2Cl2 K+ and NH3 Options: Ion-Ion , Ion-Dipole , Dipole-Dipole ,

Hydrogen Bonding, Dipole-Dipole & Ion-Dipole Forces: Strong Intermolecular Forces - Video & Lesson Transcript | Study.com

.png)

.png)